From traditional to modern compliance: How life sciences companies are making quality agile and compliant.
The Life Sciences & MedTech industries are undergoing a significant digital transformation. Technologies such as data mining and AI are reshaping development processes at their core. Many companies in these sectors focus on deep domain expertise and rely on experienced partners for areas such as big data, artificial intelligence and digital transformation. Combined with strict, country specific regulatory demands and the drive for automation, organisations face major challenges.
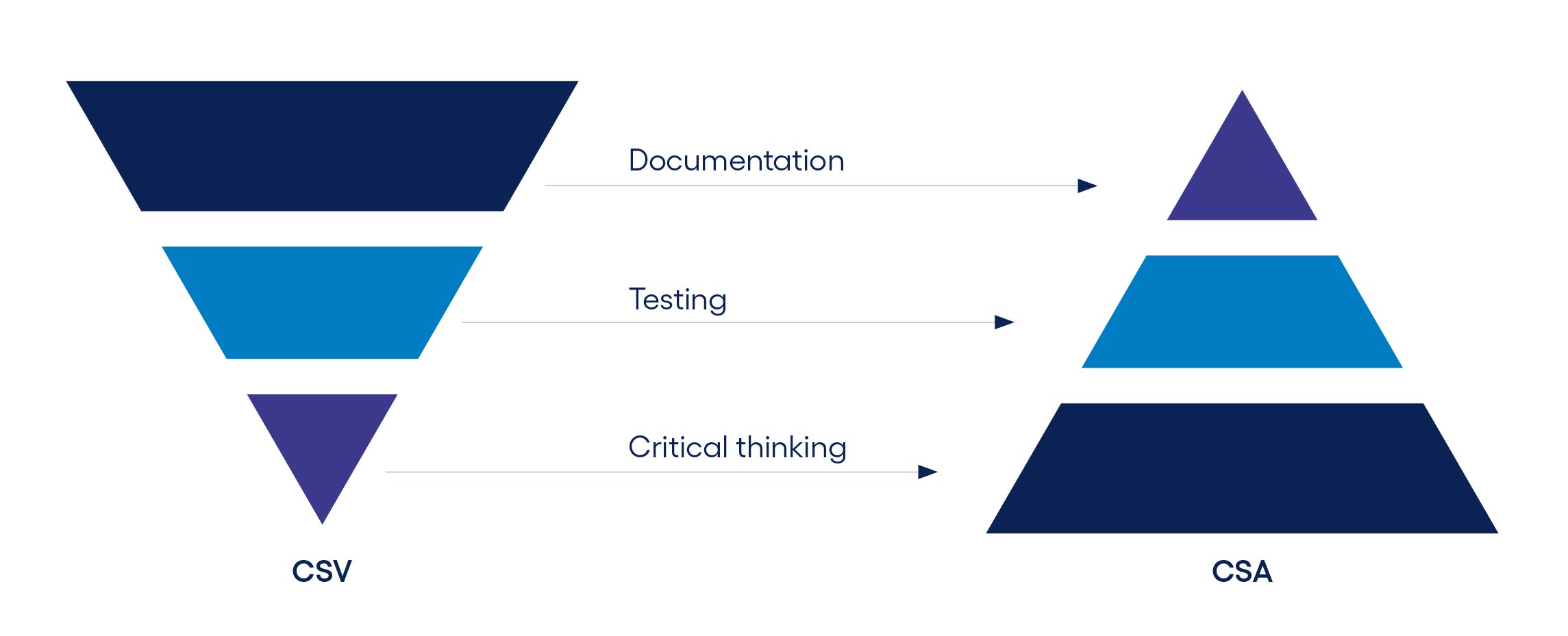
One of the biggest shifts is in how development is managed. Traditional waterfall models, where projects are planned from start to finish in advance, are increasingly outdated. Quality assurance is no longer a final stage, but an integral part of development. As a result, these processes must adapt to new realities. This is particularly evident in quality assurance: the focus is shifting away from excessive documentation of every detail, towards what matters most – critical development stages.
But how can modern development methods ensure full regulatory compliance, which remains essential for true quality in pharma and MedTech?
“Modern approaches to quality and compliance aren’t driven by paperwork - they’re driven by priorities. With the help of technology and AI, teams can focus on what truly matters: patient safety and critical risks.“
Anupama G. Nair, EDL – QE&A, Cognizant
To support agile methods without slowing them down, quality assurance must evolve. That means moving away from rigid legacy processes and embracing adaptive models. Smaller and mid-sized companies often lead the way here, as they benefit from short decision making cycles and can respond to market changes with greater agility.